Snowy Owl Bubo scandiacus Scientific name definitions
- VU Vulnerable
- Names (39)
- Monotypic
Denver W. Holt, Matt D. Larson, Norman E. Smith, Dave L. Evans, and David F. Parmelee
Version: 1.0 — Published March 4, 2020
Sign in to see your badges
Species names in all available languages
| Language | Common name |
|---|---|
| Albanian | Bufi i borës |
| Bulgarian | Полярна сова |
| Catalan | duc blanc |
| Chinese (SIM) | 雪鸮 |
| Croatian | bijela sova |
| Czech | sovice sněžní |
| Danish | Sneugle |
| Dutch | Sneeuwuil |
| English | Snowy Owl |
| English (United States) | Snowy Owl |
| Faroese | Snjóugla |
| Finnish | tunturipöllö |
| French | Harfang des neiges |
| French (France) | Harfang des neiges |
| Galician | Bufo nival |
| German | Schneeeule |
| Greek | Χιονόγλαυκα |
| Hebrew | לילית שלג |
| Hungarian | Hóbagoly |
| Icelandic | Snæugla |
| Japanese | シロフクロウ |
| Korean | 흰올빼미 |
| Latvian | Baltā pūce |
| Lithuanian | Baltoji pelėda |
| Mongolian | Цэвдгийн ууль |
| Norwegian | snøugle |
| Persian | بوف برفی |
| Polish | sowa śnieżna |
| Portuguese (Portugal) | Coruja-das-neves |
| Romanian | Bufniță de zăpadă |
| Russian | Белая сова |
| Serbian | Snežna sova |
| Slovak | belaňa tundrová |
| Slovenian | Snežna sova |
| Spanish | Búho Nival |
| Spanish (Spain) | Búho nival |
| Swedish | fjälluggla |
| Turkish | Kar Baykuşu |
| Ukrainian | Сова біла |
Bubo scandiacus (Linnaeus, 1758)
PROTONYM:
Strix scandiaca
Linnaeus, 1758. Systema Naturæ per Regna Tria Naturæ, Secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis. Tomus I. Editio decima, reformata 1, p.92.
TYPE LOCALITY:
'Habitat in Alpibus Lapponiae' ex Fn. Suec, Aves no. 46, there based on a drawing by Rudbeck.
SOURCE:
Avibase, 2023
Definitions
- BUBO
- bubo
- scandiaca / scandiacus
The Key to Scientific Names
Legend Overview
UPPERCASE: current genus
Uppercase first letter: generic synonym
● and ● See: generic homonyms
lowercase: species and subspecies
●: early names, variants, misspellings
‡: extinct
†: type species
Gr.: ancient Greek
L.: Latin
<: derived from
syn: synonym of
/: separates historical and modern geographic names
ex: based on
TL: type locality
OD: original diagnosis (genus) or original description (species)
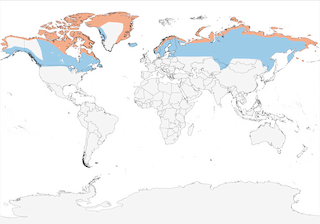
- Year-round
- Migration
- Breeding
- Non-Breeding
Distribution of the Snowy Owl




























